Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(15):5641-5652. doi:10.7150/ijbs.76699 This issue Cite
Research Paper
Traditional Chinese Medicine JingYinGuBiao Formula Therapy Improves the Negative Conversion Rate of SARS-CoV2 in Patients with Mild COVID-19
1. Department of Hepatopathy, Shuguang Hospital, Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China.
2. Medical Department, Shuguang Hospital, Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China.
3. Nursing Department, Shuguang Hospital, Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China.
4. Division of Gastroenterology and Hepatology, Department of Medicine, Indiana University, USA.
5. National Monitoring Center for Medical Services Quality of TCM Hospital, Shanghai, China.
6. Clinical Laboratory, Shuguang Hospital, Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China.
7. General Affairs Department, Shuguang Hospital, Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China.
8. Respiratory Department, Shuguang Hospital, Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China.
9. Division of Service supervision of Traditional Chinese Medicine, Shanghai Municipal Health Commission, Shanghai, China.
10. Laboratory of Cellular Immunity, Institute of Clinical Immunology, Shuguang Hospital, Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China.
11. Institute of Infectious diseases of integrated traditional Chinese and Western medicine, Shanghai, China.
# Bowu Chen and Peihua Geng contributed equally to this work.
Abstract
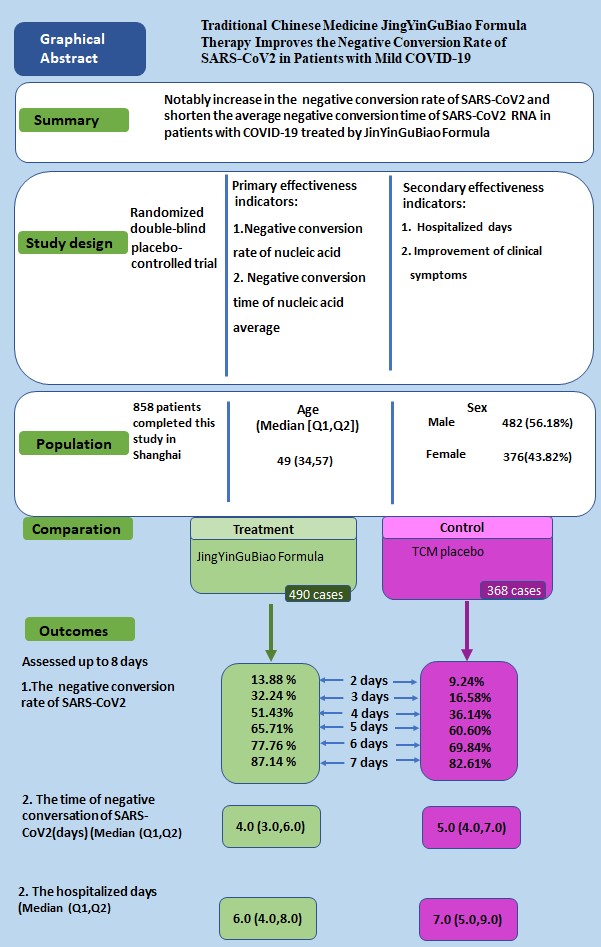
Background: Traditional Chinese Medicine (TCM) JingYinGuBiao formula (JYGB) was recommended by the Expert consensus on Traditional Chinese Medicine diagnosis and treatment of COVID-19 infection in Shanghai. We evaluated the safety and efficacy of JYGB in treating mild COVID-19 patients.
Methods: A prospective, double-blind, randomized, controlled trial was conducted (ClinicalTrial.gov registration number: ChiCTR2200058695). A total of 885 patients were randomized into the treatment group (administration of JYGB,n=508) or the control group (administration of TCM placebo, n=377) with 7-day treatment. The primary outcomes were the negative conversion rate and negative conversion time of SARS-CoV2 RNA. Secondary outcomes included the hospitalized days and symptom improvement.
Results: A total of 490 and 368 patients in the treatment and control groups completed the study. The cumulative negative conversion rates at 2 days, 3 days, 4 days, and 6 days post randomization in the treatment group were all markedly higher than those in the control group (13.88% vs. 9.24%, P=0.04; 32.24% vs. 16.58%, P<0.001; 51.43% vs. 36.14%, P <0.001; 77.76% vs. 69.84%, P=0.008). Compared with the control group, after JYGB treatment, the median negative conversion time (4.0 [3.0-6.0] vs. 5.0 [4.0-7.0] days, P<0.001) and hospitalized days (6.0 [4.0-8.0] vs. 7.0 [5.0-9.0] days, P<0.001) were reduced. While the symptoms were improved, there were no significant differences in symptom disappearance rates between both groups. In addition, further sub-group analysis showed that for patients with interval time ≤4 days or patients≤ 60 years, the clinical effects of JYGB were more remarkable with an increase in cumulative negative conversion rates, a decrease in negative conversion time and hospitalized days. JYGB was well tolerated without any severe side effects.
Conclusion: JYGB, a TCM prescription, improves the negative conversion rate of SARS-CoV2 in mild COVID-19 patients.
Keywords: COVID-19, traditional Chinese medicine, JingYinGuBiao formula, negative conversion rate, negative conversion time

 Global reach, higher impact
Global reach, higher impact